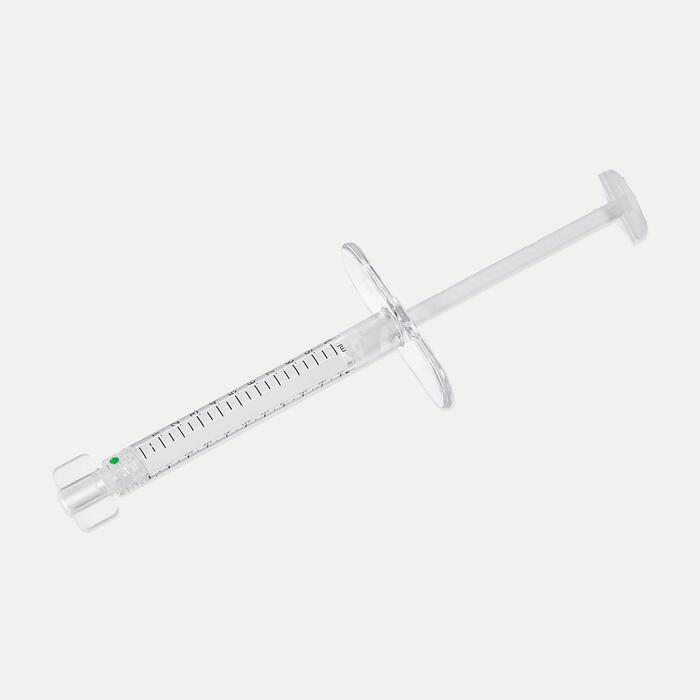
Sidekick™Sidekick™ Rigid Needle
Product Number: M0068903040
GTIN: 08714729819530
Select Product:
Sign in
orrequest an account
to view pricing and product availability
You will get an email upon availability
Frequently Bought Together
Product Details
- Small gauge needle designed for patient comfort1
- Circumferential needle markings to help guide placement1
1. Post-approval of CoaptiteTM in the treatment of Female Urinary Incontinence Post -market Study. Clinical Study Report Protocol #P1002185. Mertz North America, Inc. Coaptite PO40047/RO27.OSB Lead PMA Post Approval Study Report June 14, 2018.
Caution: U.S. Federal law restricts this device to sale by or on the order of a physician.
All images are the property of Boston Scientific.
All trademarks are the property of their respective owners.